Image source: Public Domain
Nucleus Biologics, a leading provider of custom cell culture solutions and manufacturing technologies for advanced therapies, announced the launch of NB-Lux™ AI, a next-generation digital ecosystem designed to streamline media and buffer manufacturing and management.
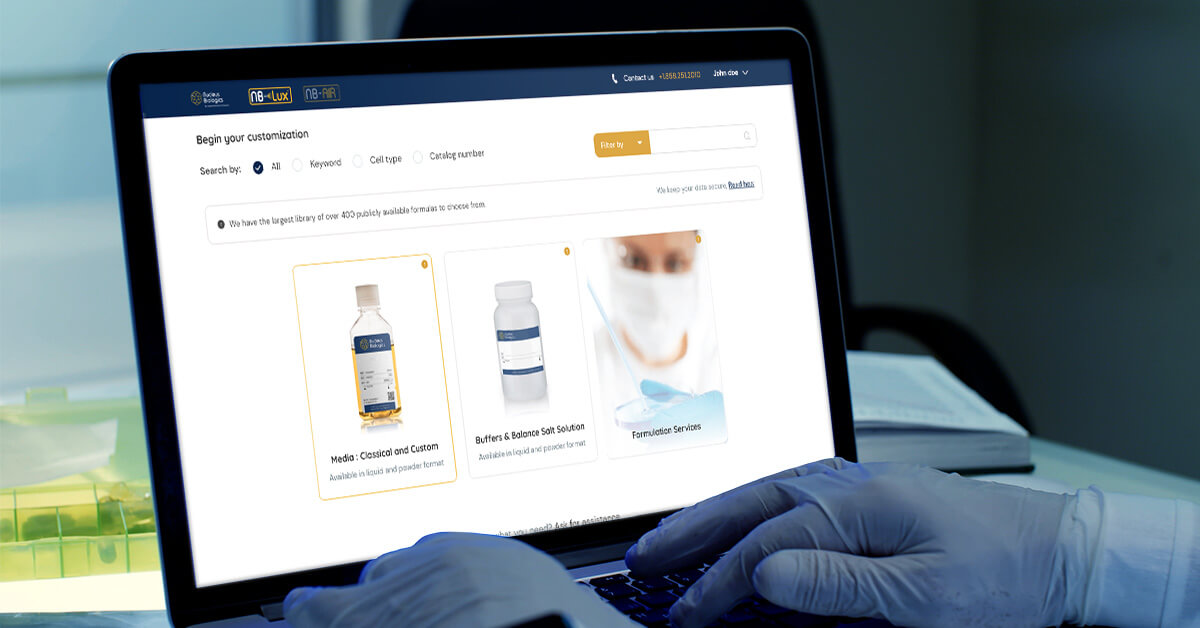
NB-Lux™, launched in 2020, was the first cloud-based ordering portal. NB-Lux™ AI builds upon that foundation, evolving into a fully integrated, AI-enabled ecosystem that eliminates manual processes and fragmented systems. This new system connects formulation design, feasibility analysis, quoting, ordering, approvals, manufacturing, and documentation into a single continuous workflow.
“Imagine an Amazon-like user experience for ordering and tracking your critical media and buffers,” said David Sheehan, Founder, President, and CEO at Nucleus Biologics. “With NB-Lux™ AI, we’ve embedded intelligence and automation across the entire process, enabling our therapy developer colleagues to receive GMP media in 4 weeks and have access to a document repository of all their lots and documents for regulatory filings.”
A Fully Digital, End-to-End Workflow
NB-Lux™ AI introduces advanced capabilities that reduce manual steps and enable faster decision-making and therapeutic development. New features include:
By digitizing the entire process, from formula to GMP release, NB-Lux™ AI enables:
Redefining Media and Buffer Sourcing
NB-Lux™ AI represents a fundamental shift in how the industry approaches media and buffer manufacturing and management, transforming a traditionally manual, fragmented process into a fully digital, connected ecosystem. With NB-Lux™ AI, developers can:
“NB-Lux™ AI is the digital backbone for modern media and buffer manufacturing,” added David Sheehan. “With today’s large language models, any scientist can get a formulation for any cell type with specific CQA’s. The next hurdle to solve is rapid GMP manufacturing. NB-Lux™ AI gets you to the clinic faster.”
By subscribing, you agree to receive email related to content and products. You unsubscribe at any time.
Copyright 2026, AI Reporter America All rights reserved.